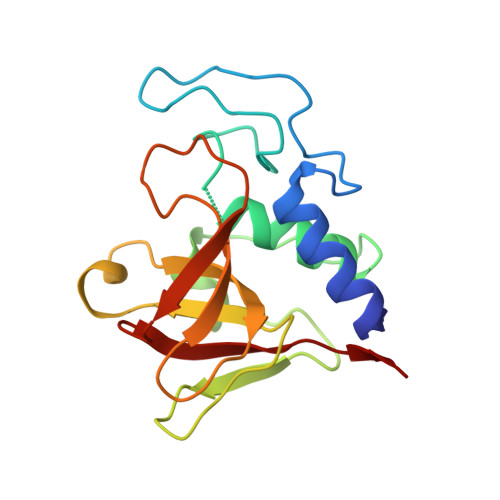
Crystal structure of the lytic CHAP(K) domain of the endolysin LysK from Staphylococcus aureus bacteriophage K.
Sanz-Gaitero, M., Keary, R., Garcia-Doval, C., Coffey, A., van Raaij, M.J.(2014) Virol J 11: 133-133
- PubMed: 25064136
- DOI: https://doi.org/10.1186/1743-422X-11-133
- Primary Citation of Related Structures:
4CSH, 4CT3 - PubMed Abstract:
Bacteriophages encode endolysins to lyse their host cell and allow escape of their progeny. Endolysins are also active against Gram-positive bacteria when applied from the outside and are thus attractive anti-bacterial agents. LysK, an endolysin from staphylococcal phage K, contains an N-terminal cysteine-histidine dependent amido-hydrolase/peptidase domain (CHAP(K)), a central amidase domain and a C-terminal SH3b cell wall-binding domain. CHAP(K) cleaves bacterial peptidoglycan between the tetra-peptide stem and the penta-glycine bridge.
Organizational Affiliation:
Departamento de Estructura de Macromoleculas, Centro Nacional de Biotecnologia (CNB-CSIC), Calle Darwin 3, E-28049 Madrid, Spain. mjvanraaij@cnb.csic.es.